The goal of my IDEA Grant project is to discover new and/or confirm experimentally measured physics of nucleus Copper-65 produced by the nuclear reaction between Oxygen-18 and Titanium-50.
Many people, including myself about a year ago, might read that sentence and think ok… and? What is the purpose of that? Reading these project titles used to make me think that these physicists use all of their intelligence and funding to study cool, obscure stuff that interests them instead of anything applicable to society, or really any other research field. Turns out, that is pretty far from the truth. Throughout my short time with FSU’s Nuclear Physics Department and Brookhaven National Laboratory’s (BNL) National Nuclear Data Center (NNDC), I’ve experienced the significance collaboration to low-energy nuclear physics: that the motivation of many research projects is to contribute to the large collection of knowledge regarding the properties of all known nuclei.

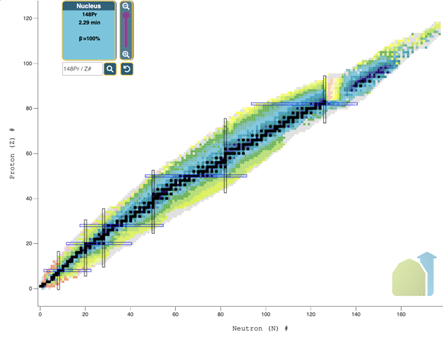
The image above is the famous chart of nuclides organized by proton (Z) number on the vertical axis and neutron (N) number on the horizontal axis. The black line going through the middle is all of the stable isotopes, i.e. show no evidence of radioactivity when it is kept in its un-excited state. The rest of the blocks (note they are the majority) in that chart undergo some type of radioactive decay: maybe more than one kind!
Each nucleus can has a unique set of excited, or energetic, states which it can be “excited” to above its ground, or un-excited, state. These excited states are organized into what’s called a level scheme. It’ll be easier to explain with an example. Let’s take a look at Copper-65’s level scheme.
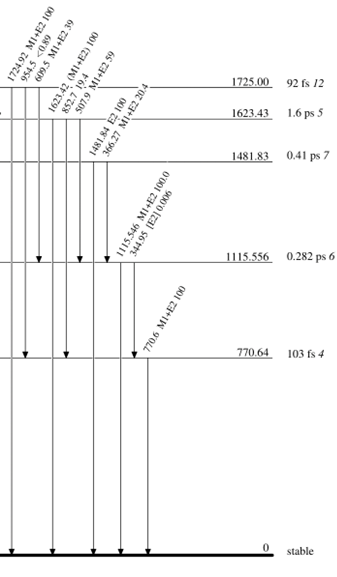
The bolded line at the bottom is the ground state and all of the levels above are excited states. The numbers to the right is the energy in kilo electron volts (keV) of each state and its half-life. The arrows represent gamma rays (labeled by their energy, angular momentum transfer, and intensity) that a nucleus may release as a means of de-exciting to its ground state.
OK. It feels like I’m going off on a tangent but stay with me. Knowing exactly how long a nucleus will stay excited for, the type of radiation it will emit, the intensity of that radiation, and how much energy that radiation could hold is the basis of all nuclear physics applications. In nuclear reactors, a neutron source is made available to Uranium-238 to form Uranium-239. Uranium-239 then undergoes fission, breaking apart into a distribution of daughter nuclei which are all in some excited state. Knowing exactly the probability of forming each daughter nuclei and the excited states they will populate helps engineers in their design of nuclear reactors to optimize energy output while prioritizing safety. Cobalt-60 works to irradiate tumors by emitting gamma-rays: knowing exactly the energy of these gamma rays and how long it will take for them to decay is crucial to developing the technology that allows for this treatment.
The two isotopes I just discussed, 239U and 60Co, were not studied with the intention of revolutionizing energy production and cancer treatment. However, after physicists discovered the capabilities of these isotopes, other scientists used their knowledge of their own respective fields which could benefit from nuclear physics, thus applying new knowledge for the benefit of society.
The goal for my research is to contribute to this large well of knowledge summarized by the chart of nuclides. One day, maybe the excited states of 65Cu end up being optimal for some other form of energy production, medicine, or technological application.
The Featured image above is a chart of Nuclides: Taken from Nudat (https://www.nndc.bnl.gov/nudat3/)