For the past couple months I have been working on my own independent project
directed toward the synthesis of a unique pattern of graphene. Possible synthetic routes
for this molecular component can be gleaned from either of the following maps which
include the formation of a scaffolding of triple bonds which the Alabugin lab is well
versed in using for unique radical cyclizations. Using this method I will start with a
simple monocyclic compound, build its high energy branching substituents, then utilize
its reactivity to form multiple cycles at once. After forming these cycles in strategic
positions I should be able to photochemically connect those new cycles forming even
more rings to result in a symmetrical, all-bay-region, form of graphene.
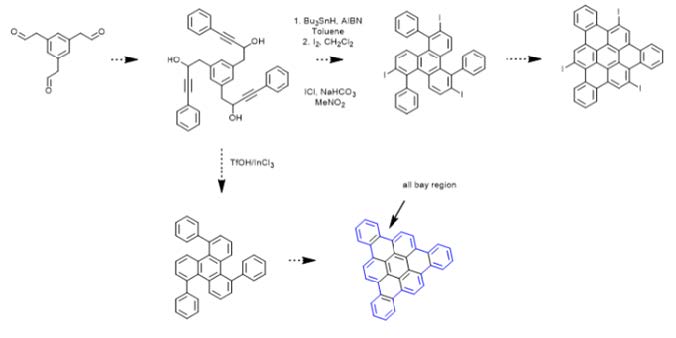
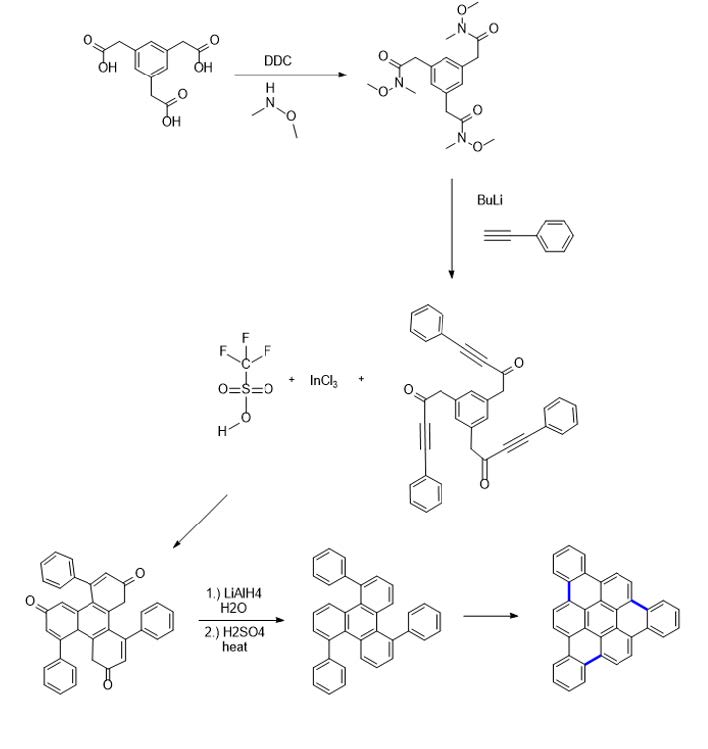
The first map’s synthesis was attempted first, although, I believe the use of a carboxylic
acid derivative such as that in the second map is another favorable alternative for future
experiments if the first route proves too challenging.
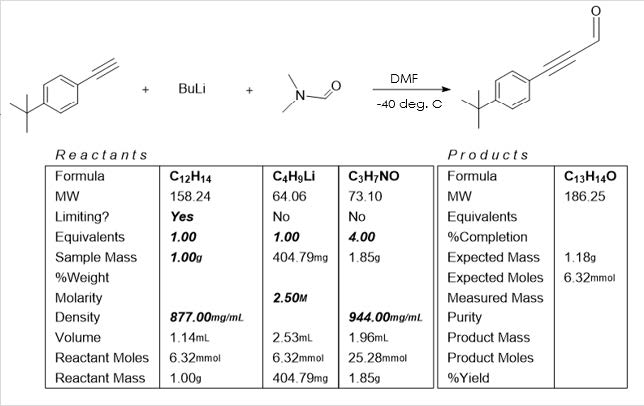
Regardless, I started with the bromination reaction below and synthesis of activated
Magnesium anthracene to prepare the tricky trisubstituted starting material necessary
for building our desired product. This also resulted in some dazzling gold solutions.

The following week, after preparing these reagents, I synthesized and purified the
desired aldehyde. Yield was surprisingly low, and NMR indicated presence of starting
material. This indicates an incomplete reaction. However, I later learned that this
reaction’s yields can be greatly increased by use of excess base, or first titrating the
BuLi reagent to determine its true concentration to assure there are sufficient moles of it
present to perform the reaction fully. This modification will be employed when this
reaction is repeated.


After a few days of purification using flash column chromatography, I was able to obtain
a very pure sample of aldehyde for the following steps. I combined this reagent with the
grignard components to attempt the formation of the trisubstituted alcohol intermediate.
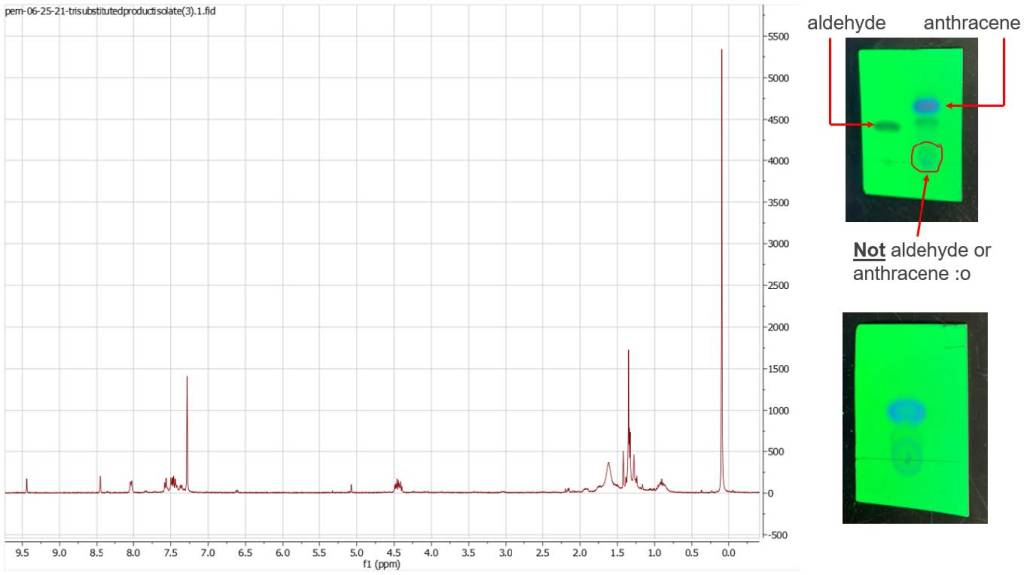
Many days were spent trying to isolate what products resulted from the reaction. The
separation proved difficult. Multiple flash column chromatography separations were
performed to only meager results; and the reaction was attempted twice on a small
scale. However, NMR data of the reaction mixture and isolated fractions of the
separations, as well as TLC analysis, indicated what could possibly be the desired
product.

Signals are weak in the nmr due to what looks like an incomplete reaction with many
starting materials left over. However, the appearance of signals in the regions between
those peaks expected for starting materials indicates the presence of what may be the
trisubstituted alcohol I am trying to obtain. This is my progress so far. Moving forward,
considering the nature of the reactions in this particular synthesis, it may be possible to
optimize and increase my yields by increasing the scale of my reactions from 1 gram to
perhaps 10 grams. Another helpful option may be to attempt these reactions with
monosubstituted starting materials, such as a simpler aldehyde or carboxylic acid,
which should give much fewer undesired side products. Finally, another very interesting
and appealing alternative route would be the use of the Barbier reaction, which uses
Zinc instead of Magnesium and can be done more simply and may yield better results.
Altogether, progress this summer has been motivating and study should continue. If the
formation of the desired graphene subunit is obtained, there is much potential in its
study. Edge modification of such molecules is highly valued, for instance, since every
unique pattern of graphene holds a unique reactivity; and the substitution patterns on
each type of edge open more and more doors for synthesis and material science.